Study explores effects of dietary choline deficiency on neurologic, system-wide health
Choline, an essential nutrient produced in small amounts in the liver and found in foods including eggs, broccoli, beans, meat and poultry, is a vital ingredient for human health. A new study explores how deficiency in dietary choline adversely affects the body and may be a missing piece in the puzzle of Alzheimer’s disease.
It’s estimated that more than 90% of Americans are not meeting the recommended daily intake of choline. The current research, conducted in mice, suggests that dietary choline deficiency can have profound negative effects on the heart, liver and other organs.
Lack of adequate choline is also linked with profound changes in the brain associated with Alzheimer’s disease. These include pathologies implicated in the development of two classic hallmarks of the illness: amyloid plaques, which aggregate in the intercellular spaces between neurons, and tau tangles, which condense within the bodies of neurons.
The new research, led by scientists at Arizona State University, describes pathologies in normal mice deprived of dietary choline as well as choline-deficient transgenic mice, which already exhibit symptoms associated with the disease. In both cases, dietary choline deficiency results in liver damage, enlargement of the heart and neurologic alterations in the AD mice typically accompanying Alzheimer’s disease, including increased levels of plaque-forming amyloid-beta protein and disease-linked alterations in the tau protein.
Further, the study illustrates that choline deficiency in mice causes significant weight gain, alterations in glucose metabolism, which are tied to conditions such as diabetes, and deficits in motor skills.
In the case of humans, “it's a twofold problem,” according to Ramon Velazquez, senior author of the study and assistant professor with the ASU-Banner Neurodegenerative Disease Research Center (NDRC) and ASU's School of Life Sciences. “First, people don't reach the adequate daily intake of choline established by the Institute of Medicine in 1998. And secondly, there is vast literature showing that the recommended daily intake amounts are not optimal for brain-related functions."
The research highlights a constellation of physical and neurological changes linked to choline deficiency. Sufficient choline in the diet reduces levels of the amino acid homocysteine, which has been recognized as a neurotoxin contributing to neurodegeneration and is important for mediating functions such as learning and memory, through the production of acetylcholine.
The growing awareness of choline’s importance should encourage all adults to ensure proper choline intake. This is particularly true for those on plant-based diets, which may be low in naturally occurring choline, given that foods high in choline are eggs, meats and poultry.
Plant-based, choline-rich foods, including soybeans, Brussels sprouts and toast, can help boost choline in these cases. Also, inexpensive, over-the-counter choline supplements are encouraged to ensure system-wide health and guard the brain from the effects of neurodegeneration.
Brain-boosting nutrient
Choline is needed to produce acetylcholine, a neurotransmitter that plays an essential role in memory, muscle control and mood. Choline also builds cell membranes and helps regulate gene expression. The established recommendations set forth by the Institute of Medicine were based on evidence preventing fatty liver disease in men. New lines of evidence imply that the established recommended daily intake of dietary choline for adult women (425mg/day) and adult men (550mg/day) may not be optimal for proper brain health and cognition. Additionally, about 90% of Americans are not meeting the recommendation levels and may not even be aware that dietary choline is required on a daily basis.

Co-lead authors of the new study Nikhil Dave (left) and Jessica Judd are researchers in the ASU-Banner Neurodegenerative Disease Research Center.
Despite decades of research and billions of dollars invested since the discovery of the devastating ailment over a century ago, there remains no therapy capable of slowing the advance of the illness. Yet, new research findings suggest that environmental and lifestyle changes, including adequate choline, may help protect the brain from Alzheimer’s disease and improve overall health.
Velazquez is joined on the study by co-lead authors and NDRC researchers Nikhil Dave and Jessica Judd. The work is highly interdisciplinary and includes researchers from the ASU Biosciences Mass Spectrometry Facility and the Translational Cardiovascular Research Center at the University of Arizona College of Medicine in Phoenix.
“This collaborative work, spanning multiple institutions and surveying the molecular processes of aging at the systems level, adds to the body of evidence produced around the importance of dietary choline in healthy aging,” said Dave.
“What I found particularly compelling about this project was that multiple organs, whose malfunction can have implications for brain health, were negatively impacted by a choline-deficient diet," said Judd.
The research appears in the current issue of the journal Aging Cell.
A prolific and mysterious killer
Alzheimer’s disease is the leading cause of dementia and the fifth leading cause of death among Americans aged 65 and older. Today, Alzheimer’s affects 6.5 million in the U.S. alone and is projected to strike close to 14 million Americans by 2060. By this time, the costs of managing Alzheimer’s are expected to exceed $20 trillion, threatening the health care infrastructure while causing immense suffering.
The accumulation of sticky protein fragments outside neurons, which form amyloid-beta plaques, and the buildup of an abnormal form of the protein tau within the bodies of neurons (tau tangles) have long been recognized signposts of Alzheimer’s disease. These brain alterations are typically followed by neurodegeneration, involving the damage and destruction of neurons. Plaques are believed to damage cell-to-cell communications in the brain while tangles block the transport of vital nutrients essential for proper cell function and survival.
In addition to amyloid-beta plaques and neurofibrillary tangles, the disease causes cell death in the brain and increasing cognitive impairment. The current work also found dysregulation of proteins in the hippocampus, a key structure affected in Alzheimer’s disease, tied to learning and memory. Both normal and AD mice showed dysregulated proteins in the hippocampus with a choline-deficient diet, with the AD model showing severe effects.
Gathering storm
The recent, dramatic increase in Alzheimer’s disease is of grave concern. Although deaths from stroke, heart disease and HIV decreased between 2000 and 2019, deaths from Alzheimer’s increased more than 145%. In addition to the toll on patients, Alzheimer’s has placed an immense burden on those caring for them. In 2021 alone, 16 billion hours of care were provided by over 11 million family members and other unpaid caregivers.
Many factors contribute to the development of Alzheimer’s disease, from genetic predisposition to age, lifestyle and environmental influences. For reasons that remain murky, females face an increased risk of developing the disease.
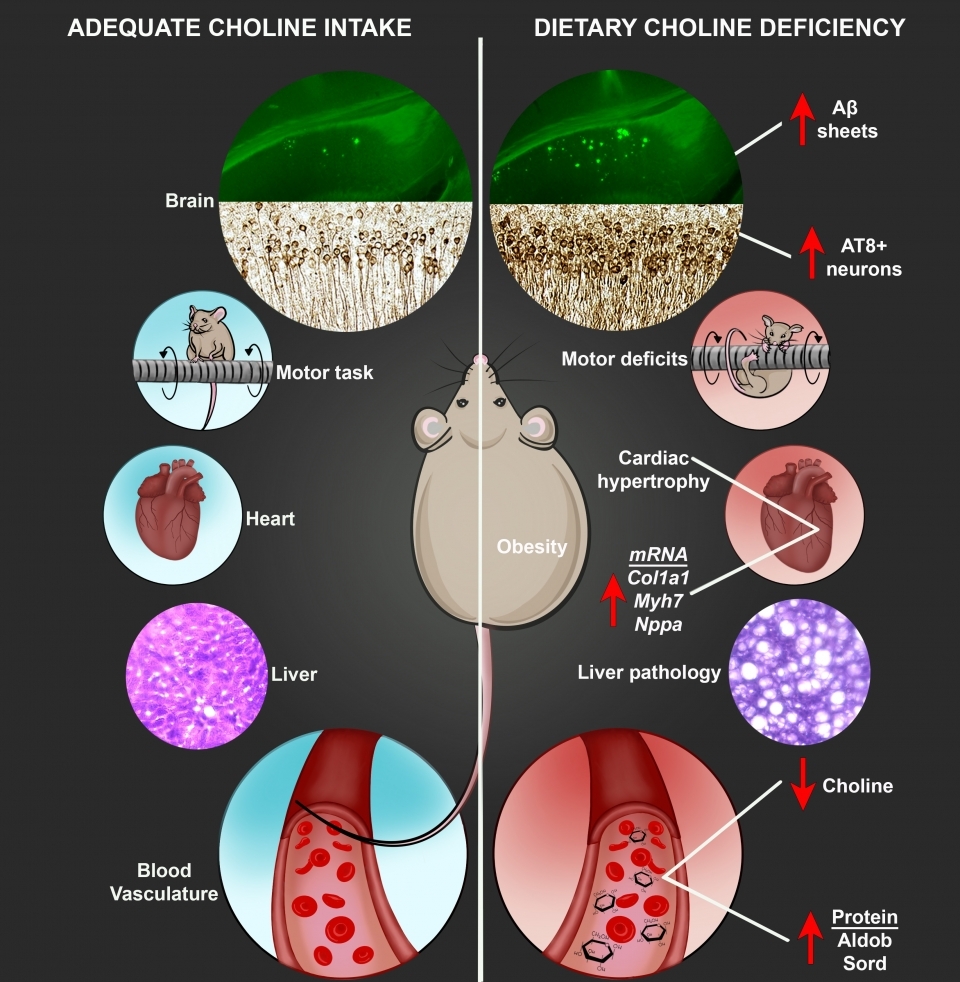
The new study highlights the effects of low dietary choline on the brain and other organs in mice. Low choline in the diet is associated with obesity, liver pathology and enlargement of the heart. Choline deficiency was also shown to be correlated with pathologies associated with Alzheimer's disease, and the low-choline mice performed poorly in tests of motor skills. Graphic courtesy the Velazquez Lab
Recent studies have identified diet as a significant factor associated with preventing cognitive decline. In earlier research, Velazquez and his colleagues demonstrated that when mice were fed a high-choline diet, their offspring showed improvements in spatial memory, compared with a normal choline regimen in the womb. Intriguingly, the beneficial effects of choline supplementation are transgenerational, not only protecting mice receiving choline supplementation during gestation and lactation, but also the subsequent offspring of these mice, suggesting inherited modifications in their genes.
Subsequent studies in the Velazquez Lab showed that choline administered to female mice throughout life yielded improvements in spatial memory, compared with those receiving a normal choline regimen.
Multifaceted effects
The new study examines mice at 3–12 months, or early-to-late adulthood, roughly equivalent to 20–60 years of age for humans. In the case of both normal and transgenic mice displaying symptoms of Alzheimer’s, those exposed to a deficient choline diet exhibited weight gain and adverse effects to their metabolism. Damage to the liver was observed through tissue analysis, as well as enlargement of the heart. Elevated soluble, oligomeric and insoluble amyloid-beta protein were detected as well as modifications to the tau protein characteristic of those leading to neurofibrillary tangles in the brain.
Further, choline-deficient mice performed poorly in a test of motor skills when compared with mice receiving adequate choline in their diet. These adverse effects were heightened in the transgenic mice. Translating these findings to humans, this implies that people who are predisposed to Alzheimer’s disease or in the throes of the illness should ensure they are getting enough choline.
The study also involved a detailed exploration of proteins in the hippocampus, an area of the brain acutely affected by Alzheimer’s disease, as well as proteins detected in blood. Dietary choline deficiency altered important hippocampal networks. These pathologies include disruption of pathways associated with microtubule function and postsynaptic membrane regulation — both essential for proper brain function. In blood, proteins produced in the liver that play a role in metabolic function were particularly dysregulated with the choline-deficient diet.
“Our work provides further support that dietary choline should be consumed on a daily basis given the need throughout the body,” Velazquez says.
Ultimately, controlled human clinical trials will be essential for establishing the effectiveness and appropriate dosages of choline, before encouraging lifelong choline supplementation. Nevertheless, the powerful new findings offer hope that choline may be one tool in the arsenal needed to defend the brain from neurodegeneration and age-related cognitive decline.
